Gallbladder Cancer (GBC) is the most common malignancy of the biliary tract which accounts for 80-95% of biliary tract cancers worldwide [1]. It is a highly malignant neoplasm with variable incidence depending on gender and geographic distribution. Risk factors include age > 60 years, female sex, obesity, chronic cholecystitis, cholelithiasis, chronic salmonella infection of gallbladder, congenital biliary tract anomalies, and a genetic predisposition. Women are affected two to six times more often than men. Gallstone is one of the main risk factors of gallbladder cancer, being present in most (~85%) patients [2,3]. In most instances, gallbladder cancer starts as metaplasia and progresses to dysplasia, carcinoma in situ and then invasive cancer. Tumour progression is very rapid and silent, resulting in poor outcome. A satisfactory outcome depends on an early diagnosis and surgical resection [3,4]. Although, tumour stage is probably the most important prognostic factor for the patient outcome, tumour infiltration and differentiation degree are also important independent prognostic factors in gallbladder cancer [2-4]. A better understanding of pathogenesis and clinicopathological characteristic of gallbladder cancer may provide insight for the development of potential diagnostic markers for this lethal disease.
Materials and Methods
In present study, a total 93 cases of gallbladder lesion were selected both prospectively and retrospectively from archives of Department of Pathology, King George’s Medical University, Lucknow, Uttar Pradesh, India. H&E sections were examined and histological grading and typing was done according to WHO classification [17]. Out of total 93 cases, 57 cases were of gallbladder carcinoma (adenocarcinoma), 27 cases of premalignant condition including 13 dysplasia and 14 metaplasia and 9 cases of chronic cholecystitis (as control). Predominant population was females (77 cases) as compared to males (16 cases) and the overall age range was 25 to 70 years (49.39±10.9 years). The cases with histopathologic diagnosis of primary adenocarcinoma, metaplasia, dysplasia and chronic cholecystitis of gallbladder were included and cases with histology other than adenocarcinoma, secondary carcinoma of gallbladder (metastatic), post chemotherapy and post radiotherapy were excluded. Distribution of cases according to gender is summarised in [Table/Fig-1].
Distribution of gallbladder lesions according to gender.
| Gallbladder lesions | Females | Males |
|---|
| n | % | n | % |
|---|
| Malignant (n=57) | 47 | 82.4% | 10 | 17.6% |
| Premalignant (Metaplasia and Dysplasia) (n=27) | 23 | 85.2% | 04 | 14.8% |
| Chronic cholecystitis (n=09) | 07 | 77.8% | 02 | 22.2% |
| Total cases (n= 93) | 77 | 82.8% | 16 | 17.2% |
The malignant cases (n=57) were further categorised according to histological grade and typing into well (18), moderately (22) and poorly differentiated (17). Different histological types were adenocarcinoma Not Otherwise Specified (NOS) (32), Papillary carcinoma (12), intestinal (3), mucinous adenocarcinoma (3), signet ring cell carcinoma (5), adenosquamous (1) and clear cell carcinoma (1).
A 3-4μ thin section was obtained from formalin fixed paraffin embedded tissue blocks and was submitted for deparaffinisation and dehydration. Immunohistochemical staining was performed using mouse monoclonal anti-human CDX2 (manufactured by Dako, FLEX; Clone DAK-CDX2 ready to use) as per manufacturer’s protocol. For the interpretation of IHC, only nuclear staining was considered positive. The percentage of immunostained tumour cells was determined semi-quantitatively by assessing the whole section and classified into four groups according to Kang GH et al.,: 0=(0% positive cells), 1=(with <10% positive cells), 2=(10–50% positive cells) and 3=(>50% positive cells) [6].
The intensity of staining was graded as 0=(absent), 1=(weak) or 2=(strong). The scores from each section were added together and a total score greater than 2 was designated as a positive result.
Statistical Analysis
Statistical analysis was done using SPSS software version 21.0. The data were summarised as number (n), percentages (%) and mean±SD (standard deviation) for each group. Quantitative variables were compared using Unpaired t-test/Mann-Whitney test (when the data sets were not normally distributed) between two groups and ANOVA/Kruskal-Wallis test (for non parametric data) between three groups. Qualitative variables were compared using Chi-square test/Fisher’s exact test as appropriate. Univariate and multivariate binary logistic regression analyses were done to assess independent predictors against dependent parameter. Disease specific overall survival analyses were determined and compared using the Kaplan-Meier method and the log-rank test. A p-value of <0.05 will be considered statistically significant.
Results
Results of Present Study are Summarised as Follows
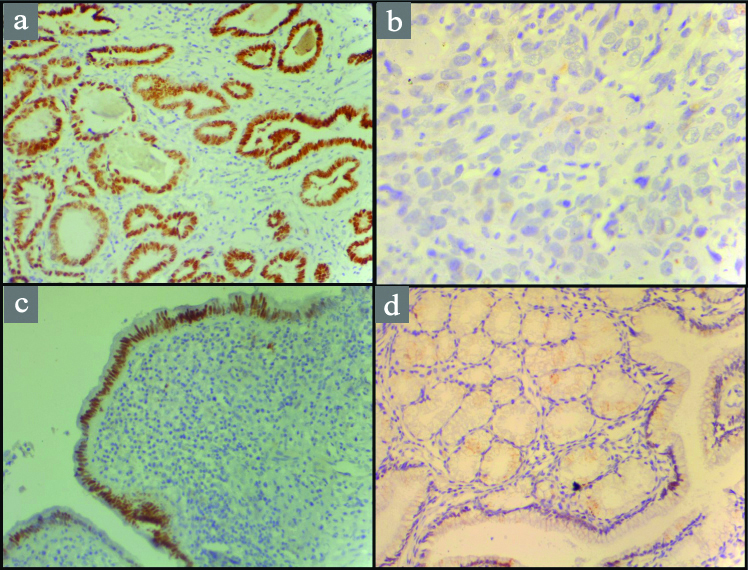
1. CDX2 expression in different histological groups: The immunohistochemistry results for CDX2 expression in different groups of malignant and premalignant lesions are summarised in [Table/Fig-2,3]. CDX2 expression was strong with well differentiated tumour and very low or lost in poorly differentiated tumour. It was also observed that CDX2 expression was Stronger in dysplasia than metaplasia cases. Among metaplasia it was more strongly associated in intestinal metaplasia (75%) than antral metaplasia (10%).
Expression of CDX2 in various histological grades of gallbladder carcinoma and premalignant lesions.
| Group | Sub Group | CDX2 expression | χ2 value | p-value |
|---|
| Positive | Negative | Total |
|---|
| Malignantlesions gallbladder (n=57) | Well differentiated | 18 (100.0%) | 0 (0.0%) | 18 (100.0%) | 18.6 | <0.001* |
| Moderately differentiated | 17 (77.3%) | 05 (22.7%) | 22 (100.0%) |
| Poorly differentiated | 06 (35.3%) | 11 (64.7%) | 17 (100.0%) |
| Premalignantlesions gallbladder (n=27) | Dysplasia | 11 (84.6%) | 02 (15.4%) | 13 (100.0%) | 8.57 | 0.003* |
| Metaplasia | 04 (28.6%) | 10 (71.4%) | 14 (100.0%) |
* Significant
CDX2 protein expression in gallbladder lesions: a) Well differentiated adenocarcinoma showing strong nuclear positivity (10X); b) Poorly differentiated adenocarcinoma showing loss of CDX2 expression (40X); c) Dysplastic mucosa showing strong nuclear positivity (10X); d) Metaplasia of gallbladder mucosa showing focal nuclear CDX2 positivity (10X) .
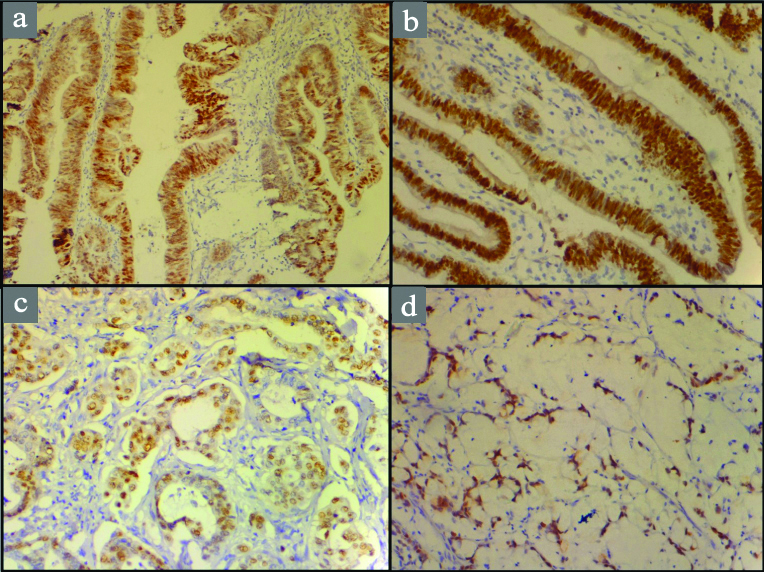
CDX2 expression in various histological types of gallbladder adenocarcinoma showed that papillary, intestinal and clear cell type showed strong association with CDX2 expression by showing 100% positivity in all the cases [Table/Fig-4,5]. Findings were statistically significant (p=0.050).
IHC positivity rate in different histological types of gallbladder carcinoma.
χ2 value=12.59; p=0.050
| Histological typing | Total | CDX2 Interpretation |
|---|
| Positive | Negative |
|---|
| NOS | 32 | 20 (62.5%) | 12 (37.5%) |
| Papillary | 12 | 12 (100%) | 0 (0.0%) |
| Intestinal | 03 | 03 (100%) | 0 (0.0%) |
| Mucinous | 03 | 01 (33.3%) | 2 (66.7%) |
| Signet | 05 | 04 (80%) | 1 (20.0%) |
| Adenosquamous | 01 | 0 (0.0%) | 1 (100%) |
| Clear cell | 01 | 01 (100%) | 0 (0.0%) |
| Total | 57 | 41 (71.9%) | 16 (28.1%) |
CDX2 protein expression in various histological types of adenocarcinoma gallbladder: a) Papillary adenocarcinoma (10X); b) Intestinal type adenocarcinoma (20X); c) Mucinous adenocarcinoma (20X); d) Signet ring adenocarcinoma (10X).
Correlation between Clinicopathological Factors and CDX2 Expression
On correlating various clinicopathological factors in malignant group, we found no correlation of CDX2 expression with age and sex, the gross tumour diameter and gallstones. In lymph node metastasis cases, we observed less expression of CDX2 (50.0%) as compare to no lymph node metastasis have stronger expression (82.05%) (p=0.012). CDX2 expression in cases with no surrounding tissue invasion came to be more strongly associated than with no surrounding tissue invasion and findings came to be statistically significant (p=0.004) [Table/Fig-6].
CDX2 Positivity rate in different clinicopathological factors.
| Clinicopathological feature | CDX2 Interpretation | χ2 value | p-value |
|---|
| Positiven (%) | Negativen (%) | Totaln (%) |
|---|
| Sex | Male | 7 (70.0%) | 3 (30.0%) | 10 (100%) | 0.884 | 0.881# |
| Female | 34 (72.4%) | 13 (27.6%) | 47 (100%) |
| Age Criteria | ≤ 50 | 25 (65.8%) | 13 (34.2%) | 38 (100%) | 2.13 | 0.145# |
| > 50 | 16 (84.2%) | 03 (15.8%) | 19 (100%) |
| Gallstones | Yes | 27 (77.2%) | 08 (22.8%) | 35 (100%) | 1.22 | 0.269# |
| No | 14 (63.6%) | 08 (36.4%) | 22 (100%) |
| Lymph Node Metastasis | Yes | 09 (50.0%) | 09 (50.0%) | 18 (100%) | 6.27 | 0.012* |
| No | 32 (82.05%) | 7 (17.9%) | 39 (100%) |
| Tumour Diameter | <2.0 cm | 06 (85.7%) | 01 (14.3%) | 07 (100%) | 0.696 | 0.706# |
| >2.0 cm | 32 (72.73%) | 12 (27.27%) | 44 (100%) |
| Grossly not identified | 04 (66.7%) | 02 (33.3%) | 06 (100%) |
| Surrounding Tissue invasion | Yes | 13 (54.16%) | 11 (45.84%) | 24 (100%) | 8.14 | 0.004* |
| No | 29 (87.9%) | 04 (12.1%) | 33 (100%) |
*Significant, #Insignificant
CDX2 Expression in Terms of Mean Survival of the Patients
For this, mean survival of the patients was calculated which came to be 8.74±5.920 months with range of one month to 24 months. After surgical resection of adenocarcinoma cases (n=57) only 37 cases were available for regular follow-up of which 23 cases were survived more than nine months with CDX2 positive rate of 87% (20/23) whereas 14 died within nine months with CDX2 positive rate of 50% (7/14) findings statistically significant (p=0.014) [Table/Fig-7].
CDX2 positivity rate and survival.
| Survival | CDX2 Interpretation | χ2value | p-value |
|---|
| Number | Positive | Negative |
|---|
| ≥ 9 months | 23 | 20 (87.0%) | 3 (13.0%) | 6.027 | 0.014* |
| < 9 months | 14 | 7 (50.0%) | 7 (50.0%) |
*Significant
Survival Graph
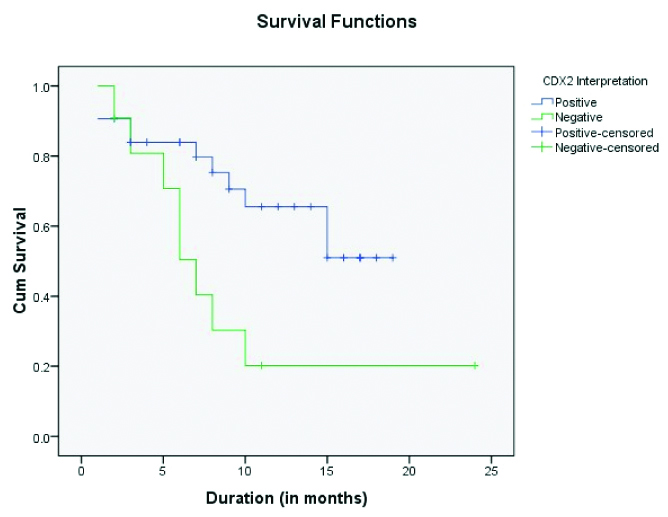
Kaplan-Meier plots [Table/Fig-8] for overall survival in 37 patients with gallbladder adenocarcinoma in relation to CDX2 expression was made and observed that positive or increased CDX2 expression is associated with increased overall survival with significant p-value (p=0.021, log rank test).
Kaplan-Meier plots for overall survival of patients with gallbladder adenocarcinoma in relation to CDX2 expression.
On univariate evaluation, among different clinicopathological factors, only lymph node metastasis and surrounding tissue invasion were found to be significantly associated with CDX2 positivity. There were 49 cases in which both lymph node metastasis and surrounding tissue invasion were done. Hence, a binary logistic regression was done [Table/Fig-9]. The outcome of binary logistic regression showed that both the variables were significantly associated with CDX2 positivity failed to show a significant association with CDX2 positivity in multivariate simulation.
Outcome of binary logistic regression (n=49).
| SN | Variable | OR | 95% CI | p-value |
|---|
| 1. | Lymph node metastasis | 0.628 | 0.135-2.901 | 0.549 |
| 2. | Surrounding tissue invasion | 0.268 | 0.062-1.159 | 0.268 |
| 3. | Constant | 6.073 |
Discussion
Gallbladder carcinoma accounts for nearly two-third of the biliary tract cancers, and the fifth most common cancer of the gastrointestinal tract [1,2]. Due to non-specific symptoms and rapid spread of the tumour, detection is often late, resulting into very poor outcome. Approximately 85% of gallbladder cancers belong to adenocarcinomas often well or moderately differentiated and have favourable prognosis as compared to 15% of squamous, adenosquamous or undifferentiated carcinoma [2-4,16].
We observed that mean age for malignant lesions was 48.81±10.21 years, for dysplasia 53.08±10.316 years and for metaplasia 48.36±9.320 years. Martinez-Guzman G et al., reported that the mean age of patients with low and high grade dysplasia, carcinoma in situ and invasive carcinoma was 42, 48, 53 and 61 years respectively [18]. In our study, the youngest patient reported malignancy was of 25-year-old, so the mean age of malignancy was lower.
In present study, CDX2 expression was not detected in chronic cholecystitis cases (0/9). Our finding was consistent with Kang GH et al., they also observed no CDX2 expression in normal gallbladder epithelium [6]. Among premalignant lesions; 11 dysplasia (84.6%) and only 4 metaplasia, (28.6%) cases showed positive CDX2 expression. Hong SM et al., found that CDX2 was expressed in 5 out of 6 dysplasia’s and three cases were positive for both CDX2 and MUC2, which further supports importance of dysplasia in gallbladder carcinogenesis [13]. Wu XS et al., found CDX2 and MUC2 expression in 3 out of 4 gallbladder cancer cell lines at the mRNA level by RT- PCR method [16]. They also studied MUC2 and CDX2 in 68 gallbladder carcinomas by the immuonohistochemistry method and observed that CDX2 was absent in the normal gallbladder epithelium but was expressed in metaplasia, dysplasia and 36.8% gallbladder carcinomas. Well differentiated carcinomas had high CDX2 expression 54.8% as compared to moderately differentiated 7.1% and poorly differentiated carcinomas 0.0% [19].
In present study, CDX2 expression was present in 71.9% cases of adenocarcinoma. It showed inverse relation between CDX2 expression and tumour grade. CDX2 was strongly expressed in well differentiated carcinomas 100% followed by moderately differentiated carcinomas 77.3% and least expression in poorly differentiated carcinomas 35.3% positivity in tumour cells. Kang GH et al., found that CDX2 was expressed more frequently in well (7/7, 100.0%) differentiated adenocarcinomas than in moderately (2/6, 33.3%) and poorly (1/5, 20.0%) differentiated types [6]. Li QL et al., reported that among well differentiated 55.2%, moderately 44.8% and poorly differentiated 23.3% showed positive association with CDX2 expression and concluded that CDX2 was strongly relevant to grades of tumour [19]. Chang YT et al., reported that, CDX2 expression in Well differentiated was11/38 (28.9%), Moderately 15/71 (21.1%) and Poorly 1/28 (03.5%) with no statistical significance (p=0.07) to tumour grade or stage in between CDX2 positive or CDX2 negative cases [12].
Further we studied the histological variants of gallbladder carcinoma and classified them according to the WHO classification [17,20]. We observed that Papillary adenocarcinoma and Intestinal type adenocarcinoma of gallbladder show 100% CDX2 expression (12/12 and 3/3 cases respectively). Although Signet ring cell carcinomas were assigned as WHO Grade-3 tumour they showed strong association, that was 4/5 (80%) cases showed CDX2 expression.
Among NOS adenocarcinoma (not otherwise specified), 20/32 (62.5%) showed CDX2 expression. Mucinous adenocarcinoma was showed poor CDX2 expression that was 1/3 (33.3%).
In accordance to this Kang GH et al., observed CDX2 expression in 10/18 adenocarcinomas, not otherwise specified, 9/9 papillary adenocarcinomas and 1/1 intestinal-type adenocarcinoma [6]. Li QL et al., studied 11 cases of mucinous carcinoma and found that 5/11 cases (45.5%) showed positive association with CDX2 expression [19].
Correlation between CDX2 Expression and Clinicopat-hological Factors
On correlating the association of CDX2 with various clinical and pathological factors we found that lymph node metastasis and surrounding tissue invasion showed statistically significant association with CDX2 expression. It was higher (82.05%) in cases with no lymph node metastasis in contrast to lymph node metastasis (50.0%). It was also found than gallbladder carcinoma with surrounding tissue invasion showed lower expression CDX2 (54.16%) as compared to no surrounding tissue invasion (87.9%).
Park JS et al., did a study on 38 cases of Stage II gallbladder cancer and found that infiltrating and poorly differentiated types were independent prognostic factors of recurrence after curative resection for Stage II gallbladder carcinoma [21]. Our finding is supported by Li QL et al., whom stated that CDX2 and Hep expression was an independent predictor of survival in addition to lymph node status and surrounding tissue invasion at the time of diagnosis [19]. They reported negative correlation (p < 0.01 or p < 0.05) between CDX2 or Hep expression and tumour size, lymph node metastasis and surrounding tissue invasion.
CDX2 Expression in Terms of Survival
In present study for survival analysis the mean survival time came to 8.74±5.920 months, and observed that 23 patients survived more than 9 months showed high CDX2 expression 87% in contrast to 50% expression in short survived, 14 patients who died within nine months post surgery. We also found that CDX2 expression was more strongly associated with better prognosis of the patients of gallbladder carcinoma. This finding was supported by Chang YT et al., they found that CDX2 alone was as independent predictor of survival after resection of Biliary Tract Cholangiocarcinoma (BTC) [12]. CDX2 and tumour stage were independent prognostic factor in patients with biliary tract carcinomas. Hong SM et al., reported that patients with both CDX2 and MUC2 expressing extrahepatic BTC had a better overall survival in univariate but not multivariate analysis than patients with other tumours [13]. CDX2 expression has been considered as one of the good prognostic markers in patients with gastric carcinoma [8,11,22], pancreatic tumour [23], and carcinoma of the ampulla of Vater [14].
In our study, the relevance of positive CDX2 expression to patient’s survival was examined by Kaplan-Meier survival analysis. It showed overall survival has positive association with increased expression frequencies of CDX2 (p=0.021).
Limitation
The limitations of our study were small sample size, several procedural differences, postoperative care and chemotherapy. Further studies on a larger sample size might help in establishing the usefulness of various clinicopathological factors associations in a multivariate scenario.
Conclusion
CDX2 protein expression has inverse relation with tumour grade, its expression is minimum or absent in Grade 3 and 4 tumours. CDX2 is an independent marker of clinical outcome in addition to lymph node metastasis and surrounding tissue invasion in gallbladder adenocarcinoma patients. It can be used as prognostic marker for gallbladder carcinoma.