Septicemia has been one of the most common causes of death in intensive care unit (ICU) patient. The accurate and timely diagnosis of such cases is extremely important for proper patient management. However, with the overlapping clinical signs and symptoms with those of other acute critical medical conditions, achieving a high level of certainty while diagnosing septicemia can be a difficult challenge in which the laboratory has a crucial role in providing invaluable information. The tests most often ordered in this scenario are the complete blood count with manual differential & blood culture [1].
The blood culture is considered the “gold standard” investigation for diagnosing septicemia. But it has its own short comings such as low sensitivity, the need for sterile collection techniques to avoid contamination and false positivity. Usually there is a delay of at least a few days before results of blood culture are available, leading the clinicians to initiate empirical antibiotic therapy that must be withdrawn & a sensitive antibiotic needs to be added later. This whole process often leads to antibiotic resistance [1,2]. CRP, IL-6 & procalcitonin levels are the other markers which have their own sensitivity and specificity in predicting sepsis [1,3,4].
Review of peripheral blood smears can also yield important diagnostic information through the identification of the morphologic changes characteristically seen in reactive neutrophils & monocytes during infection. Such changes include the presence of toxic granulation, toxic vacuolization and Döhle bodies in the cytoplasm. Immature forms (left shift), such as bands and metamyelocytes, can also be identified. Similarly monocytes will show an increase in the cell size and presence of granules in the cytoplasm. Examining the smears is quite arduous & time staking approach. Other problems encountered in peripheral smear examination are the differential count may not be precise as only 100 to 200 cells are counted, variation in identification of leukocytes among the different observers and uneven distribution of cells in the smear for counting. Hence peripheral smear has its own merits & demerits in the accurate diagnosis of sepsis [1,2].
Materials and Methods
This prospective case-control study was done for a period of one year, i.e., from June 2013 to May 2014 where in peripheral blood samples from 94 patients with infections were studied using the VCS parameters by the Coulter ® LH 780 Haematology analyser. Among the 94 patients with infection, 36 patients had systemic infection and 58 patients had localized infections. The criterion for systemic infection was positive blood culture. Patients with localized infections presented as organ-specific infections like pneumonia, urinary tract infection, acute pyelonephritis, and broncho-pulmonary infections. Clinical signs (temperature > 38.5oC and purulent sputum) and positive results on chest radiography were the basis for diagnosing pneumonia and broncho-pulmonary infections. Clinical signs and positive results for urinary culture of significant pathogens defined urinary tract infection and acute pyelonephritis. Forty six (46) control samples were selected randomly from among cases with CBC count and differential data within normal limits {WBC count, <11,000/μl (<11.0 × 109/L); percentage of neutrophils, <85% (0.85)} and without signs of infection. The patients’ medical records were also reviewed and the clinical details were retrieved for clinical correlation. Ethics committee clearance was obtained for the study from the institutional ethics committee.
The data collected includes the total WBC count, the mean channels of neutrophil volume (MNV), neutrophil conductivity (MNC), neutrophil light scatter (MNS) and mean channels of monocyte volume (MNV), monocyte conductivity (MNC) and monocyte light scatter (MNS) obtained from the Coulter ® LH 780 Haematology analyser (Beckman Coulter. Inc). Manual differential count was not performed.
Statistical Analysis
All analysis were performed using IBM SPSS Statistics 20.0 software. Results were expressed as the mean ± standard deviation (SD). Comparisons between mean values of the groups were performed by ANOVA and subsequent post-hoc range tests, and multiple comparisons were performed in pairs to determine which mean values differ. A two-tailed p-value < 0.05 was considered statistically significant. The diagnostic properties of each test were investigated by receiver operator characteristic (ROC) curves. The optimum cut-off value for each variable was tested. Subsequently, the sensitivity, specificity, and area under the ROC curve were assessed.
Results
Age and distribution of sex are illustrated in [Table/Fig-1]. The data from 94 patients with infections (systemic infections; positive blood culture for bacteria n=36 and localized infections n=58) were analysed using VCS parameters. The most common bacteria cultured was Staphylococcus aureus (24 cases), followed by Escherichia coli (6 cases), Bacillus (2 cases), and Klebsiella, Proteus, Enterococcus, Streptococcus pneumoniae (one each).
Baseline characteristics, VCS values of Neutrophils & Monocytes
| Systemic infection (SI) | Localised infection (LI) | Control (C) | SI vs LI | SI vs C | LI vs C |
|---|
| Number | 36 | 58 | 46 | | | |
| Age (yrs) | 72.6 | 69.2 | 65.2 | - | - | - |
| Sex (M:F) | 1.2:1 | 1:1.4 | 1.3:1 | - | - | - |
| WBC (x105/ml) | 14.6 | 24.5 | 7.7 | <0.001 | 0.008 | <0.001 |
| VCS parameters |
| Neutrophils |
| Volume (MNV) | 160.5 | 156.8 | 137.2 | 0.402 | <0.001 | <0.001 |
| Conductivity (MNC) | 145.3 | 146.5 | 143.3 | NS | NS | NS |
| Scatter (MNS) | 138.8 | 138.3 | 147.2 | NS | <0.001 | <0.001 |
| Monocytes |
| Volume (MMV) | 183 | 174.5 | 161.7 | 0.01 | <0.001 | <0.001 |
| Conductivity (MMC) | 125.5 | 125.9 | 119.3 | NS | <0.001 | <0.001 |
| Scatter (MMS) | 89.5 | 86.7 | 87.7 | NS | NS | NS |
p-value < 0.05 is considered significant
The WBC count ranged from 11,000/μl (11 × 109/L) to 59,600/μl (59.6 × 109/L); mean, 24,537/μl (24.5 × 109/L) in the localised infection group & 4,000/μl (4 × 109/L) to 48,000/μl (48 × 109/L); mean, 14,650/μl (14.6 × 109/L) in the systemic infection group. In the control group, the WBC count ranged from 4300/μl (4.3 × 109/L) to 11,000/μl (11 × 109/L); mean, 7700/μl (7.7 × 109/L).
Comparison of VCS parameters in Cases vs Controls: We observed a significant increase in the MNV & MMV was observed in patients with infections (both systemic & localised) as compared with control subjects MNV: 158.3 ± 13.7 vs 137.2 ± 4.3; p<0.001) & MMV: 177.8± 16 vs 161.7 ± 6.04; p <0.001). The MNS was decreased significantly in patients with infections (138.5 ± 9.2 vs 147.2 ± 3.8; p<0.001). The MMC was increased significantly in patients with infection (125.8 ±6.0 vs 119.3 ±4.8; p<0.001).
Comparison of VCS parameters in Systemic infection vs Localised infection
When the VCS parameters of systemic infections were compared with localised infections, MMV was significantly increased in systemic infection as compared to localised infection (183 ± 19.2 vs 174 ± 12.7, p=0.01). However, there was no significant difference in the MNV of patients with systemic infection & localised infections (160.5±17.5 vs 156.8±10.5, p>0.05).
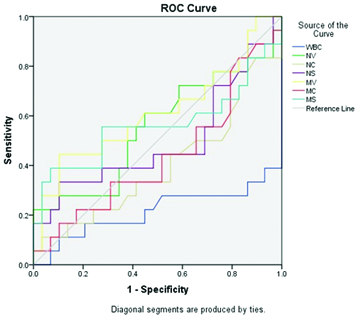
With a cutoff of 150 for the MNV, a sensitivity of 72% and specificity of 70% was achieved. Similarly, with a cut off of 170 for MMV, a sensitivity of 67% and specificity of 59% was achieved [Table/Fig-2].
Sensitivity & specificity in predicting acute bacterial infections
| Parameter | Cut off Points | Sensitivity (%) | Specificity (%) |
|---|
| MNV | >150 | 72 | 70 |
| MMV | >170 | 67 | 59 |
The ROC curve of inflammatory markers highlights the MNV & MMV as a marker of infection [Table/Fig-3].
ROC curve of inflammatory markers highlighting mean neutrophil volume (MNV) & mean monocyte volume (MMV)
Discussion
In routine practice, the WBC count, absolute neutrophil count, percentage of bands and immature neutrophils has been used to predict bacterial infections. A detailed examination of morphological changes in neutrophils such as toxic granules, cytoplasmic vacuoles and Dohle bodies will further provide important diagnostic information. Advent of automated analysers such as Coulter® technology has revolutionized the laboratory medicine practice, making the diagnosis quick and time saving. In the present study, the clinical usefulness of VCS parameters was determined in the early diagnosis of systemic bacterial infections. The parameters studied such as MNV, MNS, MNC, MMV, MMC & MMS are evaluated for each of the samples run through the coulter, without additional sample requirement nor is it time consuming. It as well reduces the stress on the pathologists who has to evaluate thousands of cells to look for the reactive features [4].
In the present study, we established that the morphologic changes seen in left-shifted neutrophils and reactive monocytes during an episode of acute bacterial infection could be measured quantitatively using the Coulter ® LH 780. The VCS parameters, MNV & MMV were seen to be significantly elevated and MNS significantly decreased in samples from patients with infection. MNV and MMV are sensitive and reliable indicators of infection in our body [Table/Fig-2]. However, MMV was seen to be significantly increased in patients with systemic infections as compared to localised infections, suggesting the possible use of this parameter as an indicator of acute systemic infection and to differentiate it from localised infection.
Since the advent of the coulter ® analyser, few studies have been carried out to analyse the clinical utility of the VCS technology for early diagnosis of septicaemia and bacterial infections. Studies comparing the VCS parameters of neutrophils showed that the MNV was significantly raised and MNS was significantly reduced in sepsis patients as compared to controls [1,6,7]. Similarly other studies found a significant increase in the MNV & MMV among the patients with sepsis as compared to localised infections as well as control [2,4]. Some authors investigated the value of Neutrophil Distribution Width (NDW) and demonstrated that, compared to controls; the neutrophil population of patients with infection is less homogeneous, with a wider variation of individual cell sizes, as shown by the increased NDW [7–9]. In a study by Celik et al., a significant increase in MNV, and significant decrease in MNC and MNS was observed in septic newborns [5] [Table/Fig-4].
Comparison among different studies of the VCS parameters in predicting acute infection
| MNV | Sensitivity (%) | Specificity (%) | MNS | Sensitivity (%) | Specificity (%) | MMV | Sensitivity (%) | Specificity (%) |
|---|
| Chaves et al., [1] | >145 | 83 | 54 | <141 | 46 | 80 | - | - | - |
| Mardi et al., [2] | >150 | 76 | 63 | - | - | - | >170 | 86 | 40 |
| Celik et al., [5] | >157 | 79 | 82 | <127.5 | 60 | 65 | - | - | - |
| Present study | >150 | 72 | 70 | | | | >170 | 67 | 59 |
We found a significant increase only in MMV among the patients with systemic infections as compared to localised infections suggesting the specificity of MMV for diagnosing systemic infections. A significant increase in MNV values on Coulter ® analysis was observed in both systemic as well as localized infections, however this parameter was found to be statistically insignificant in differentiating the cause as systemic or localized. Hence, MMV could be taken as an early indicator of systemic infection and MNV as an indicator of any bacterial infection in the body. This was in contrast to the study by Mardi et al., where they found a significant increase in MNV as well as MMV [2].
The present study is prospective, and the smaller sample sizes studied thus far may have introduced bias despite of all care. There may have been incorporation bias, overestimating the diagnostic accuracy of the investigated markers. VCS parameters can offer several advantages in clinical settings but large prospective cohort studies are required to validate the clinical usefulness in other causes of neutrophilic leukocytosis. These causes may include the increased release from bone marrow stores owing to tissue infarction or ischemia (hypoxia), decreased margination owing to exercise or epinephrine medication, stress, decreased extravasation into tissue due to use of glucocorticoids, and increased numbers of bone marrow precursors owing to chronic inflammation, tumours, or myeloproliferative disorders and granulocyte-colony stimulating factor stimulation [1,4]. Further, clinical scores related to organ failure and outcome such as Acute Physiology and Chronic Health (APACHE) score should be used to evaluate the patient group quantitatively [4]. The VCS parameters of leukocytes can also be used to evaluate different types of febrile illness such as dengue, malaria & tropical frebrile illness [10].
Conclusion
Sepsis which still remains a critical clinical issue despite of advances in its diagnosis requires a rapid but yet an accurate tool for its early detection. Though blood culture is considered the gold standard, much more easily and quickly obtained VCS parameters from automated analysers has emerged as a promising Haematologic finding for diagnosis of sepsis. Through our study, we believe that MNV and MMV have the potential to be used as early indicators of acute non-systemic & systemic bacterial infections; we would like to highlight MMV as an emerging predictor of acute systemic infections.
p-value < 0.05 is considered significant