Post-operative pain is inevitable following major surgery, resulting from surgical trauma and handling of tissues [1]. The inadequate relief of post-operative pain has adverse physiological effects resulting in delay of patient recovery and return to daily activities [2]. Caesarean delivery patients have even more compelling reasons to achieve optimal post-operative pain relief than other surgical patients since inadequate pain control impairs breastfeeding and delays maternal-infant bonding. It is usually relieved by opioids or Non-Steroidal Anti-Inflammatory Drugs (NSAIDs) but with an additional challenge that the drug used should be safe during lactation.
Tramadol, a centrally acting analgesic has opioid and non-opioid modes of action, both acting synergistically [3]. It has been shown to provide effective analgesia after both intra-muscular and intravenous routes of administration in post-operative pain [1,4]. NSAIDs are also used for post-operative pain relief and commonly used are diclofenac, ketorolac, piroxicam, lornoxicam, selective cyclooxygenase (COX-2) inhibitors like etoricoxib and parecoxib [5]. Few studies have been carried out to compare the efficacy and tolerability of opioid and NSAID especially tramadol versus piroxicam for management of post-operative pain [5,6]. Both these drugs have been reported to be safe during lactation and hence, the choice, even though more novel analgesics are available whose safety remains a concern. Hence, this study was undertaken to compare the efficacy and safety of tramadol and piroxicam in the treatment of post-caesarean section pain in primigravidae.
Materials and Methods
A prospective interventional, open label, parallel group study was conducted by Departments of Pharmacology and Obstetrics and Gynaecology. Pregnant women admitted to the Department of Obstetrics and Gynaecology were included. The study was carried out from January 2012 to December 2013. Protocol designed for the study was approved by the Institutional Ethics Committee and written informed consent was obtained from women willing to participate in the study.
A total of 120 primigravidae women aged between 20-35 years, scheduled for elective caesarean section under spinal anaesthesia were enrolled into the study. They were assigned to two groups of 60 women each. Group 1 received single dose of injection piroxicam 20mg and group 2 injection tramadol hydrochloride 100mg intra-muscularly immediately after recovery from anaesthesia. Women with history of allergy to piroxicam and tramadol, pre-eclampsia, eclampsia, peptic ulcer, gastrointestinal bleeding, asthma, Chronic Obstructive Pulmonary Disease (COPD), hemorrhagic disorders, renal and hepatic dysfunction were excluded from the study.
Pain intensity was measured using Visual Analogue Scale (VAS) which is graded from 0 to 10 (“0” = no pain and “10” = worst pain). VAS score is classified as painless (0), mild (1-4), moderate (5-8), and severe (9-10) [5,7]. It was explained and administered at baseline (before administration of the drugs) and at 2, 4, 8, 12 and 24 hours post-operatively. Rescue analgesic, injection butorphanol 2mg was intended to be administered intra-muscularly to patients if VAS score was more than four during post-operative period (either at 2, 4, 8 or 12 hours) [8].
Pulse rate, blood pressure and respiratory rate were monitored immediately after recovery from anaesthesia and at 2, 4, 8, 12 and 24 hours post-operatively. Sedation was scored using five point scale with zero being alert, 1– sedated, 2 – drowsy, 3 – asleep and 4 – comatose [9]. Sedation was assessed at baseline, two and four hours post-operatively. Adverse events if any were recorded. Patient’s satisfaction was assessed at the end of 12 hours on a four point scale with score of 1 as poor, 2 fair, 3 good and 4 excellent [10].
Statistical Analysis
To detect the mean difference in VAS score 2.9 with a standard deviation of 3.2 at the end of 2 hours following drug administration, considering an α error of 0.05, power of 90% and 10% dropout rate, the sample size was calculated to be 30 in each group using two independent sample test.
Descriptive data were expressed as mean±standard deviation and analysed using descriptive statistics. Student t–test was used to analyse continuous data and intra-group analysis was done by applying repeated measure ANOVA and post-hoc Bonferroni adjustment test. Categorical data was analysed by Chi-square test. The p-value of 0.05 or less was considered statistically significant.
Results
One hundred and twenty primigravidae were included in the study. [Table/Fig-1] represents the age distribution and duration of surgery in both the groups. Primigravidae in the age group of 20-25 years were more in both the treatment groups with 78.33% in piroxicam group and 95% in tramadol group. Both the groups were comparable.
Age distribution and duration of surgery.
| Variable | Piroxicam | Tramadol |
|---|
| Age (years) | 23.32±3.43 | 22.03±2.017 |
| Duration of surgery (minutes) | 52.42±8.15 | 50.33±10.03 |
[Table/Fig-2] shows a significant reduction in mean VAS score at 2, 4, 8, 12 and 24 hours compared to baseline (p<0.001) in both the groups. At baseline, the mean VAS score was comparable between the groups. After administration of the study drugs, the mean VAS score was significantly lower at 2, 4 and 8 hours in piroxicam group compared to tramadol group. The area under curve for both the groups [Table/Fig-3,4] was calculated by trapezoid method and the pain intensity from baseline to 24 hours was less in piroxicam group (4.56) compared to tramadol group (4.87).
Comparison of mean VAS scores.
| VAS | Baseline | 2 hrs | 4 hrs | 8 hrs | 12hrs | 24hrs | †p value |
|---|
| Piroxicam | 6.65±0.82 | 2.68±0.79 | 1.67±0.72 | 1.58±0.62 | 2.58±1.38 | 2.78±0.71 | 0.001 |
| Tramadol | 6.35±0.89 | 3.03±0.63 | 2.02±1.03 | 2.22±1.34 | 2.65±1.40 | 2.95±0.72 | 0.001 |
| *p-value | 0.059 | 0.009 | 0.034 | 0.001 | 0.793 | 0.207 | |
* - unpaired ‘t’ test †- repeated measure ANOVA
Area under curve for VAS score for Piroxicam.
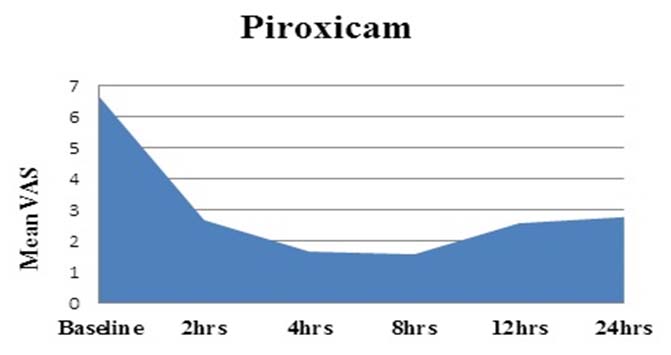
Area under curve for VAS score for Tramadol.

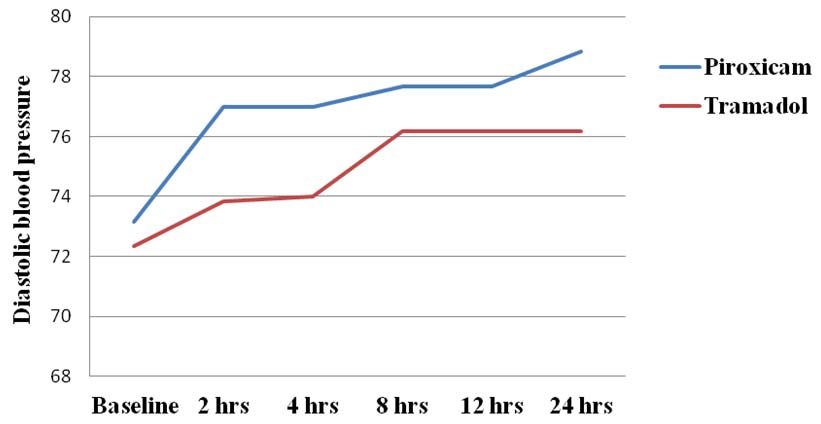
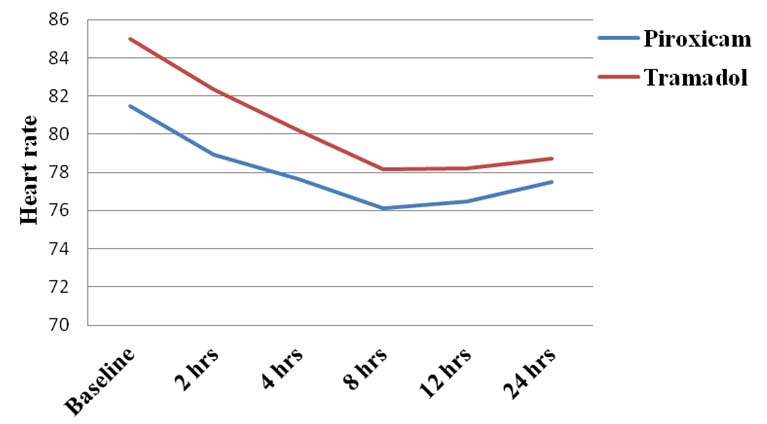
Rescue analgesic was required after 4 hours in both the groups. Total number of patients requiring rescue analgesic injection butorphanol 2mg in piroxicam group was 12 and 21 in tramadol group. Therefore, the total amount of rescue analgesic required in piroxicam and tramadol group was 24mg and 42mg respectively, as shown in [Table/Fig-5]. The systolic Blood Pressure (BP) remained within normal limits but the diastolic BP which was low at baseline started gradually increasing upto 24 hours with piroxicam compared to tramadol. There was a significant difference in diastolic BP up to 4 hours between piroxicam and tramadol groups (p<0.001) and thereafter, the difference was minimal and statistically not significant up to 24 hours as shown in [Table/Fig-6]. Heart rate was more than 80 beats/min at baseline which gradually reduced up to 8 hours and thereafter, it stabilized in both the groups. The decrease was significant at all the assessed six intervals (p<0.001) between the groups as shown in [Table/Fig-7].
Time of rescue analgesic.
| Time of rescueanalgesic inhours | Number ofpatients (%) inPiroxicam group | Number ofpatients (%) inTramadol group | p-value |
|---|
| 4 | 1 (1.67) | 3 (5) | 0.309 |
| 8 | - | 8 (13.33) | 0.003 |
| 12 | 11 (18.33) | 10 (16.67) | 0.810 |
Diastolic blood pressure in both the groups.
Heart rate in both the groups.
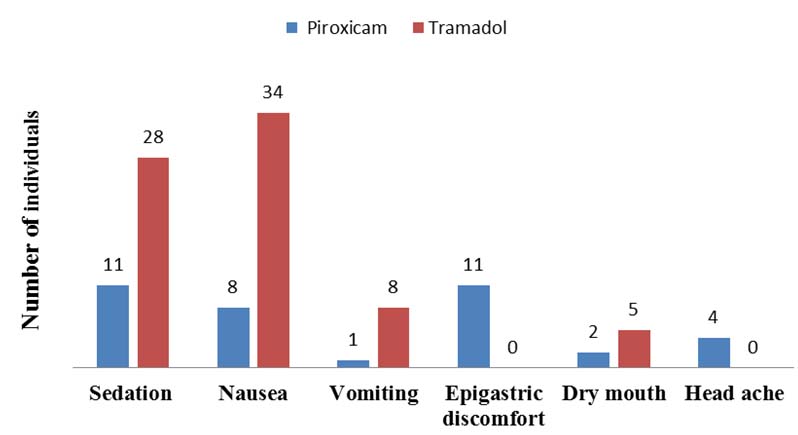
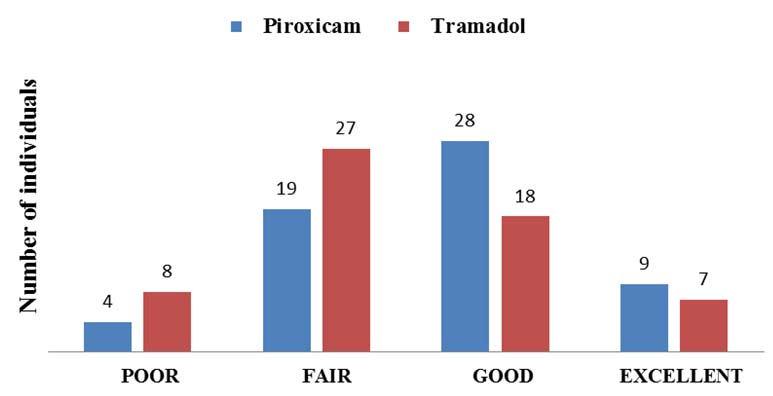
[Table/Fig-8] represents the adverse effects of both the study drugs. Eleven and 28 patients reported sedation score of ‘1’ in piroxicam and tramadol groups respectively. Sedation and nausea was significantly higher in tramadol group (p<0.001 and p=0.043 respectively) whereas the epigastric discomfort which did not warrant treatment in any women and headache was commonly observed with piroxicam (p<0.001 and p=0.042 respectively). At the end of 12 hours, 46.66% of patients graded their satisfaction score as good and 15% as excellent in piroxicam group whereas, 30% graded as good and 11.66% as excellent in tramadol group as shown in [Table/Fig-9].
Adverse effects of the study drugs.
Patient satisfaction score.
Discussion
Caesarean section is a major surgical procedure and it differs from other major laparotomies because women have to recover quickly to provide care for their new-born within few hours following surgery. Pain is the result of incision causing damage to the skin and other tissues which affects adversely the quality of life of patients and their care givers necessitating adequate control of post-operative pain [11–13]. In this study, only primigravidae were included since the intensity of pain differs in primigravidae and multigravidae. Piroxicam an NSAID of oxicam class is a potent non-selective, reversible inhibitor of COX leading to inhibition of prostaglandin synthesis which has a role in producing pain and development of inflammation [14]. Tramadol is a synthetic opioid which has spinal and supraspinal analgesia [6].
In both the treatment groups, the reduction in pain was significant at all points of time (up to 24 hours) when compared to baseline and similar findings were observed in other studies [5,6]. These findings indicate that both the study drugs are effective as analgesics for post-operative pain for a longer duration of time. In piroxicam group, pain relief was significant at 2, 4 and 8 hours post-operatively compared to tramadol group. This suggests that patients receiving piroxicam had better analgesic effect compared to tramadol. In a study conducted by Farshchi et al., there was no significant difference between the groups at any point of time in reducing post-operative pain. The mean VAS score at 12 hours in our study was 2.58±1.38 in piroxicam group which is less compared to the finding by Farshchi et al., (3.3±0.4) [5]. Gradual reduction in pain up to 12 hours was observed in both the groups but further reduction was not observed as shown in [Table/Fig-2]. This probably could be due to the weaning effect of the drugs after 12 hours but the analgesic effect lasted for 24 hours.
Yu-yan et al., showed that at the end of 24 hours the mean VAS score with epidural tramadol 100mg for post-caesarean section pain was 2.24±0.76 which was slightly lower a value compared to our study (2.95±0.72) [15]. This difference could be explained on the basis that epidural route of administration delivers the drug directly to the receptors or nerves exiting the spinal cord.
In many studies, the vital signs like blood pressure and heart rate were observed intra-operatively [16,17]. Since, they are the important indicators of hemodynamic stability even during post-operative period, they were monitored post-operatively for 24 hours in the present study. The diastolic blood pressure which was low was probably due to sympathetic blockade following spinal anaesthesia. Here in both the groups, increase in the diastolic blood pressure was observed but it was significant in piroxicam group compared to tramadol at all points of time. This could be explained by the fact that piroxicam through inhibition of COX, reduced the vasodilating prostaglandins leading to water retention and increased total peripheral resistance. It is also influenced by increased renal endothelin-1 synthesis by piroxicam [18]. The heart rate was high immediately following recovery from anaesthesia which may be due to patient’s agony with pain. Following reduction in pain intensity, heart rate gradually stabilized which was significant with piroxicam, thus, reflecting that pain relief was better with piroxicam.
The adverse effects in both the groups were well tolerated. Eleven and 28 patients reported sedation in piroxicam and tramadol groups respectively whereas in Farshchi et al., it was 22 and 35 patients in piroxicam and tramadol groups respectively [5]. Sedation, nausea and vomiting may interfere with feeding and infant care. These adverse effects due to tramadol limit its use during post-natal period. To overcome the above disadvantages, NSAIDs like diclofenac, piroxicam can be used. Epigastric discomfort was more in piroxicam group due to inhibition of protective effect of prostaglandin on gastric mucosa mediated by COX inhibition which can be adequately treated or prevented with proton pump inhibitors like omeprazole. At the end of 12 hours, 46.66% and 15% graded their satisfaction score as good and excellent respectively in piroxicam group whereas in tramadol group it was 30% and 11.66% respectively indicating that patients were contented with piroxicam therapy.
Limitation
The tolerability of intra-muscular piroxicam compared to tramadol was studied for 24 hours in our study. Multiple doses in the post-operative period beyond 24 hours may be further studied. Patients should be monitored for gastrointestinal adverse effects and level of sedation due to piroxicam and tramadol twice a day for 3-5 days.
Conclusion
Piroxicam and tramadol provided adequate pain relief after caesarean delivery but piroxicam was better compared to tramadol in terms of pain relief and tolerability. Thus, intra-muscular injection of piroxicam can be used as an alternative to tramadol for treating acute pain following caesarean section as it has better analgesic effect and devoid of central depressive effects of opioids.
* - unpaired ‘t’ test †- repeated measure ANOVA